+33 (0) 3 20 16 91 40
Statistical processing

An extensive assessment of your analytical performance
- The statistical treatment is carried out following AGLAE's procedures covered by accreditation. Its is based on normative documents such as ISO 13528 "Statistical methods for use in proficiency testing by interlaboratory comparisons" and ISO 5725 "Accuracy (trueness and precision) of measurement methods and results".
- The indicators of analytical performances (z-score mainly) are calculated by consensus of the results of participants who analysed during the recommended period to start the sample treatment.
- A guide to interpreting the test reports in chemistry and in microbiology is provided to the participants of the tests. It explains in detail the statistical processing carried out and its interpretation.
A clear and complete test report
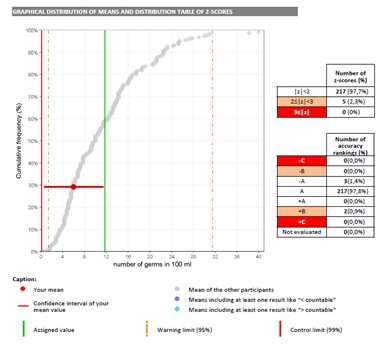
- Easy interpretation with colour codes
-
Calculation of analytical performances indicators (z-score and ranking) from consensus values
-
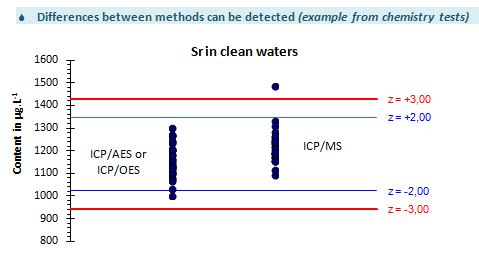
Detailed with an evaluation of the quality of test materials and with various interpretations: differences between mehods, influence of the culture media…
-
Translated into English (except test reports of the medical biology field and related to macroinvertebrates)
A personnalized report
You receive a personalized test reports to get quickly to the essential points: your results.
The first part contains the presentation of the test, the test materials quality and the possible differences between methods.
The second personalized part for a quick access to your performance information:
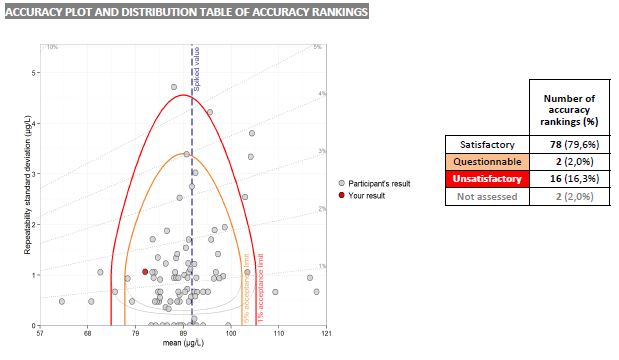
- Your results and performance for all the parameters of the test are gathered in a single table: at a glance, you identify the quality of your analyses
- The data of your laboratory and the general information of the test are summarized per parameter. In addition, your key data will be highlighted on all the graphs
To have a look at a test report, browse AGLAE's leaflet (links from page 3)
An answer to the issues of uncertainties of measurements
-
For chemistry tests: Calculation of a zeta-score which enables you to check the relevance of your uncertainty of measurements estimates. A guide to calculating your uncertainties from proficiency testing data in compliance with ISO 11352 is available from your member area to assist you in your calculations.
- For some microbiology tests, repeatability uncertainties (ur²) and reproducibility (uR²) are calculated and presented for each laboratory. These estimates enable you to quantify the technical uncertainty of analytical devices within interlaboratory repeatability and reproducibility conditions.
A summary of all your results at any time and for the period of your choice
This summary is useful for your Internal Quality Control, any applications waiting for approvals and during your audits.
- Whenever needed, participants generate from their member area an Excel file for the chemistry and microbiology tests.
- This document summarizes, for all the parameters, the precision values calculated during proficiency tests, the results participants returned and their analytical performance.
